Movex is a medicine that has chondroprotective properties. The drug is used to normalize bone and cartilage tissue. With long-term use of the medicine, it is possible to establish phosphorus-calcium metabolism, thanks to which a person stops degenerative processes.
Movex is based on glycosaminoglycans and proteoglycan - together they form a compound that, in its chemical composition, differs little from ordinary human cartilage. To achieve maximum results, long-term use of Movex is required.
pharmachologic effect
Movex is based on glucosamine, a natural building material for joints. The component accelerates the restoration processes in cartilage, protects them from damage, accelerates the regeneration process and normalizes the recovery process.
Regular use stops degenerative and pathological processes. These two substances make up the natural structure of cartilage, thanks to which it is possible to return the joints to their natural structure.
Glucosamine is a component that is a structural part of normal human cartilage tissue. When ingested, this substance promotes the accelerated production of proteoglycans. Also, such changes contribute to faster absorption of sulfates by the joints.
Proteoglycans compensate for the deficiency of useful components that are necessary for the construction of structural bone tissue. It should be borne in mind that glucosamine, which is present in the base, is responsible for the production of hyaluronic acid. Such changes help slow down degenerative processes in bone tissue.
The active components of Movex are necessary for the production of chondroitinsulfuric acid. It helps improve the quality of synovial fluid and normalizes the rate of deposition of many chemical components in the body.
Long-term use of the drug helps relieve pain caused by damage to bone tissue. Such changes in the body will only occur if you take the medicine for a long time.
Movex has a fairly powerful chondroprotective effect. This is provided by chondroitin sulfate. The active components accelerate the process of regeneration of cartilage and bone tissue and have a powerful anti-inflammatory and analgesic effect.
The drug has a positive effect on the state of phosphorus-calcium metabolism and significantly slows down the rate of bone loss. Long-term therapy helps replenish the required amount of joint fluid.
Features of good stagnation
Zagalni.
To minimize risks, the lowest effective dose of vicoristan should be followed by the shortest treatment period necessary to control symptoms.
In case of persistent NSAIDs, there is an increased risk of rupture, perforation or bleeding, especially in late-life patients.
The risk of thrombotic cardiovascular and cerebrovascular complications is increased in association with the use of selective COX-1/COX-2 inhibitors and other NSAIDs.
Remnants of the cardiovascular risk of diclofenac may grow with higher dosages and the need for treatment, which must be stagnated for a short period and at the lowest effective dose.
The neurological effects of NSAIDs include the suppression of blood pressure and/or arterial hypertension, which is why diclofenac should be used with caution in patients with impaired heart function and obviously There are other factors that lead to the shutdown of the country.
Caution is also recommended for patients who are taking diuretics or concomitant ACE inhibitors or who may have increased risk of hypovolemia.
Cardiovascular and cerebrovascular manifestations.
A clear medical opinion and consultation will be required for patients with hypertension and/or congestive heart failure of the lungs or death stage in the anamnesis, subject to stagnation of NSAIDs, including diclofenac, steroid The countryside was getting dark and full of dirt.
A slightly increased risk of arterial thrombotic events (such as myocardial infarction or stroke) may be associated with diclofenac administration, especially in high doses and during trival administration.
It is not recommended to treat patients with uncontrolled arterial hypertension, congestive heart failure, persistent ischemic heart disease, occlusive disease of peripheral arteries and/or cerebrovascular diseases. illnesses.
Patients with cardiovascular risk factors (for example, hypertension, hyperlipidemia, cellular diabetes, chicken) should be treated with diclofenac only after clinical evaluation and at a dose of ≤ 100 mg sparsely. since the bath is no more painful than just a few days. You should periodically review the patient's need for ingested diclofenac to relieve symptoms and response to therapy. Patients should be careful before serious symptoms of atherothrombosis appear (eg, chest pain, shortness of breath, weakness, loss of consciousness), which may develop without delay. Patients should be informed that in such cases it is necessary to immediately contact a doctor.
Floating on the nirk.
Remnants under the hour of stagnation of NPZD, including diclofenac, were careful to block the middle and swelling, special care should be taken in patients with impaired heart function or low blood pressure in medical history, summer age patients, patients who are undergoing concomitant treatment with diuretics or drugs that can be infused daily on the function of niro, as well as in patients with a corresponding decrease in the volume of postpartum tissue for any reason, for example, before or after surgical procedures. In such cases, when taking diclofenac, it is recommended to monitor the function of the drug. After the therapy is completed, the patients' legs usually turn to the point where they have received a bath.
Floods into the scolio-intestinal tract.
Intestinal hemorrhage (hematemesis, melena), infection or perforation, which can be fatal, has been reported with the use of non-steroidal anti-inflammatory drugs, including diclofenac. These ulcers may appear at any time during the course of treatment, regardless of the presence or presence of precursor symptoms or serious bowel ulcers in the history.
In summer-age patients, such complications may cause more serious complications. In cases of scutulo-intestinal bleeding or symptoms in patients, treatment with diclofenac should be avoided, and the drug effect will be minimal. As with all NSAIDs, close medical attention and special care are required in patients prescribed diclofenac with symptoms that indicate damage to the side of the scolio-intestinal tract, with suspected virus, bleeding whose, perforation or such conditions in the anamnesis.
The risk of bleeding, bleeding or perforation in the scolio-intestinal tract increases with increased doses of NSAIDs, including diclofenac, as well as in patients with a history of bleeding or perforation. Yes, but in patients of the summer age.
To reduce the risk of toxic influx into the grass tract, treatment should be started and maintained with the lowest effective doses.
Below is a look at the nutritional plan for combination therapy with congestive drugs (such as misoprostol or proton pump inhibitors) for such patients, as well as for patients who require concomitant congestion a bath of medicinal products to take low doses of acetylsalicylic acid (aspirin) or other medicinal products to promote rizik damages the scolio-intestinal tract.
Patients with a history of scutulointestinal toxicity, especially during the summer, will require caution for the detection of non-minor abdominal symptoms (especially scutulointestinal bleeding).
Care must be taken for patients to avoid concomitant treatment with drugs that increase the risk of irritation or bleeding, such as systemic corticosteroids, anticoagulants, such as warfarin, selective reversal inhibitors. serotonin captivity (SIDS) or antiplatelet drugs, such as acetylsalicylic acid.
Patients with virulent colitis or Crohn's disease require close medical supervision and caution when ingesting the drug, as debris may become congested.
Float onto the skin.
Very rarely, serious skin reactions, some fatal, including exfoliative dermatitis, Stevens-Johnson syndrome, and toxic epidermal dermatitis, have been reported in association with NSAIDs, including diclofenac. necrolysis. The greatest risk for the development of these reactions is at the beginning of therapy; in most cases, during the first month of treatment. You should take the drug at the first manifestations of skin viscera, swelling of the mucous membrane, or any other manifestations of hypersensitivity.
Pour onto the stove.
Close medical supervision is required in cases where the drug is prescribed to patients with impaired liver function, and fragments of their liver may be damaged. As with all NSAIDs, including diclofenac, one or more liver enzymes may be elevated. Elevations in the level of liver enzymes may not be accompanied by the appearance of clinical symptoms. Such growth is possible to a moderate level (³ 3 to < 8 times above the upper limit of the norm) or to a pronounced advance (³ 8 times above the upper limit of the norm). The growth of fermentative enzymes, as a rule, changes after the drug has been stagnated. Most of the episodes were prevented from growing to border levels.
It is recommended to monitor liver function indicators throughout the treatment course. As the improvement of normal liver function indicators is maintained or intensified, clinical symptoms of liver disease appear or other manifestations are avoided (eosinophilia, syphilia), stagnation Suvannya to the drug slid pripiniti. Hepatitis can progress within an hour of taking diclofenac without prodromal symptoms. OKRIM ZROSTANNYA RIVNIV PechINKOVKHKOVKH, RIDKO was gone about the heavy reaction of the Pechinki, including the Zhovtyanitsyu I Fulmіantnny hepatitis, the necrosis of the Pechini і Pechinkov, Yaki Inodi Buli. Use diclofenac with caution in patients with hepatic porphyria, as this may provoke an attack.
Suspension when asthma is evident.
In patients with asthma, seasonal allergic rhinitis, swelling of the nasal mucous membrane (for example, nasal polyps), chronic obstructive diseases of the lungs or chronic infections of the respiratory tract (especially if and association with symptoms similar to allergic rhinitis) reactions to NSAIDs similar to asthma exacerbation (the so-called aspirin asthma with intolerance to analgesics), angioedema and urticaria occur more frequently than in other patients. When dealing with such illnesses, special precautionary approaches are recommended (preparedness until emergency assistance is given). There are also patients who may have allergic reactions, such as hysteria, itching, or urination, in other words.
Hematological manifestations.
The medicine should be followed by a short course of treatment to relieve symptoms during acute illness. When this drug is prescribed, it is recommended (as for other NSAIDs) to regularly monitor the hemogram.
Like other NSAIDs, diclofenac may immediately inhibit platelet aggregation, so careful care should be taken in patients with impaired hemostasis, hemorrhagic disease or hematological diseases.
Masking the sign of infection.
Like other NSAIDs, diclofenac, due to its pharmacodynamic effects, can mask the symptoms of infection.
SCHV and mixed illness of healthy fabric.
Patients with systemic vulgaris (SVC) and mixed diseases of the healthy tissue of possible movements are at risk of aseptic meningitis.
Zapobizhni come in.
A trace of the uniqueness of one-hour administration of the drug and systemic non-steroidal anti-inflammatory drugs, such as selective cyclooxygenase-2 inhibitors, through the presence of any evidence of a synergistic effect and in association with potential additive side effects effects.
At the ridki vipads, Yak I, at the fast of the NPZZ, can be instructed to be instructed by the Alergіchychi reaction, the number of anathylactic/anathylacta -ore react, and the diclophenaka to the diclophenaka without ahead.
With care, administer the drug to patients over 65 years of age, especially if they are physically weakened, or if the patient’s body weight is below normal.
Barvnik zhvykh zakhid FCF (E 110) may cause allergic reactions.
Suspension during pregnancy or breastfeeding.
Women's fertility.
Like other NSAIDs, diclofenac can affect women's fertility and is therefore not recommended for women planning pregnancy. The following is a look at nutrition about the use of diclofenac for women who cannot become pregnant, as well as for women who are undergoing treatment for complete infertility.
The drug should not be used during pregnancy or breastfeeding.
This is due to the fluidity of the reaction during treatment with vehicles or other mechanisms.
Please note that when taking the drug in the recommended dose and with a short course of treatment, there is no risk of reaction. For patients treated with Movex ®
The active cause is a disruption of the function of the central nervous system, not when used by motor vehicles or when used with folding mechanisms.
Pharmacokinetics
The active components of this medicine are perfectly absorbed by the body. About 25% of glucosamine sulfate is rapidly absorbed by body tissues immediately after receiving the drug. The highest concentration of this substance is present in the kidneys, liver and cartilage tissue.
90% of glucosamine is absorbed by the small intestine and travels through the bloodstream to the liver. Under the influence of metabolites, the product breaks down to the state of urea, carbon dioxide and ordinary water.
30% of the active ingredients of the drug can be present for a long time in the connective tissues of the body. The components of the drug are excreted naturally through the kidneys and liver in the form of urine and feces.
Sodium chondroitin sulfate causes reactions in the body 3-4 hours after use, while its maximum concentration in the synovial fluid occurs after 5 hours. This component is completely eliminated by the body in just 25 hours.
Movex active tablets No. 30
Compound
active ingredients: 1 film-coated tablet contains glucosamine sulfate 500 mg, sodium chondroitin sulfate 400 mg, potassium diclofenac 50 mg
excipients: povidone, microcrystalline cellulose, colloidal silicon dioxide, talc, croscarmellose sodium, magnesium stearate, sodium starch (type A), hypromellose, polyethylene glycol 6000, titanium dioxide (E 171), sunset yellow dye FCF (E 110).
Dosage form
Film-coated tablets.
Basic physicochemical properties: film-coated tablets, orange in color, oval, biconvex, notched on one side.
Pharmacological group
Drugs affecting the musculoskeletal system. Combined anti-inflammatory drugs.
ATX code M01BX.
Pharmacological properties
The drug has anti-inflammatory, analgesic, chondroprotective and regenerative effects. Slows down the process of damage to cartilage tissue and bone resorption, restores cartilage tissue, accelerates the formation of callus in case of injury, and helps restore joint function.
Pharmacodynamics.
Glucosamine is a substrate for the construction of articular cartilage and stimulates the regeneration of cartilage tissue. Any adverse effect (disease, age-related metabolic disorders, injuries) reduces its synthesis and concentration in connective tissue, which leads to disruption of the structure and function of joints and the appearance of pain. Glycosaminoglycans and proteoglycans are part of the complex matrix that makes up cartilage.
Glucosamine is part of the endogenous glycosaminoglycans of cartilage tissue. Glucosamine sulfate has chondroprotective properties, reduces the deficiency of glycosaminoglycans in the body and is involved in the synthesis of proteoglycans and hyaluronic acid. Due to its ability to selectively act on cartilage tissue, glucosamine initiates the process of sulfur fixation during the synthesis of chondroitinsulfuric acid, normalizes calcium deposition in bone tissue, helps restore joint function and eliminate pain. Glucosamine sulfate selectively acts on articular cartilage, is a specific substrate and stimulator of the synthesis of hyaluronic acid and proteoglycans, inhibits the formation of superoxide radicals and enzymes that cause damage to cartilage tissue (collagenase and phospholipase), prevents the destructive effect of glucocorticoids on chondrocytes and the disruption of glycosaminoglycan biosynthesis caused by non-steroidal anti-inflammatory drugs them drugs.
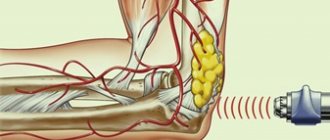
Chondroitin sulfate is a high-molecular polysaccharide involved in the construction of cartilage tissue, reduces the activity of enzymes (elastase, hyaluronidase) that destroy articular cartilage, maintains the viscosity of synovial fluid and stimulates the regeneration of articular cartilage. Affects the phosphorus-calcium metabolism of cartilage tissue, slows down bone resorption. Slows down the progression of osteoporosis. In the early stages of the inflammatory process, sodium chondroitin sulfate suppresses its activity and, thus, slows down the degeneration of cartilage tissue. Helps reduce pain, improve joint function and reduce the need for NSAIDs. Prevents compression of connective tissue, “lubricates” articular surfaces and normalizes the production of joint fluid.
Diclofenac potassium is an NSAID with analgesic, anti-inflammatory and antipyretic activity and has a rapid onset of action, which is especially important for the treatment of acute pain in inflammatory conditions. The mechanism of action is associated with a non-selective blocking of the synthesis of the enzyme cyclooxygenase, which leads to inhibition of the synthesis of prostaglandins, which play a major role in the development of inflammation, pain and fever.
Diclofenac potassium suppresses predominantly the exudation phase, to a lesser extent proliferation, reducing collagen synthesis and associated tissue sclerosis.
Reduces pain at rest and during movement, reduces the manifestations of morning stiffness in joints, swelling of soft tissues, improves the functional state of the musculoskeletal system. Pharmacokinetics.
Glucosamine sulfate. The bioavailability of glucosamine when administered orally is 25-26%. After distribution in tissues, the highest concentrations are determined in the liver, kidneys and cartilage tissue. Approximately 90% of glucosamine that enters the body orally, in the form of glucosamine salt, is absorbed into the small intestine and enters the liver through the portal bloodstream. A significant portion of glucosamine is metabolized in the liver. Breaks down to urea, water and carbon dioxide. About 30% of the dose taken persists for a long time in the connective tissue. It is excreted by the kidneys and in very small quantities in feces.
Chondroitin sulfate. After a single dose of chondroitin, the maximum concentration in the blood plasma (Cmax) is achieved after 3-4 hours, in the synovial fluid - after 4-5 hours. The concentration in synovial fluid exceeds the concentration in blood plasma. The bioavailability of chondroitin sulfate is 13-15%. Excreted by the kidneys within 24 hours.
Diclofenac potassium does NOT accumulate. The maximum concentration in blood plasma is achieved 2:00 after administration. Plasma protein binding - 99.7%. Penetrates into synovial fluid. Systemic clearance of the active substance is 263 ml/min. The half-life is 1-2 hours. 60% is excreted in the urine in the form of metabolites, less than 1% is excreted unchanged by the kidneys, and the remainder is excreted in the form of metabolites in bile.
Indications
— Treatment of diseases of the musculoskeletal system, accompanied by signs of inflammation, pain, degenerative-dystrophic changes in the cartilage tissue of the joints and spine, and decreased joint mobility.
— Osteoarthritis, periarthritis (including knee, hip joints, intervertebral osteochondrosis, spondyloarthrosis), rheumatoid arthritis.
— Fractures and injuries (to accelerate the formation of callus), post-traumatic inflammation of soft tissues and the musculoskeletal system (due to sprains, impacts).
Contraindications
Hypersensitivity to the components of the drug. Do not use if you are allergic to shellfish. Acute stomach or intestinal ulcer; history of gastrointestinal bleeding or perforation, history of gastrointestinal bleeding or perforation after the use of non-steroidal anti-inflammatory drugs (NSAIDs), history of acute or recurrent gastric or intestinal ulcer. Inflammatory bowel disease (Crohn's disease or ulcerative colitis). Contraindicated in patients who, in response to the use of acetylsalicylic acid or other NSAIDs, experience attacks of bronchial asthma, Quincke's edema, urticaria or acute rhinitis. Severe liver failure (Child-Pugh class C, cirrhosis or ascites). Severe renal failure (creatinine clearance ˂30 ml/min). Tendency to bleeding, thrombophlebitis, phenylketonuria. Congestive heart failure (NYHA II-IV). Treatment of PERIOPERATIVE pain during coronary artery bypass grafting (or use of a heart-lung machine). Ischemic heart disease in patients with angina pectoris, a history of myocardial infarction; cerebrovascular diseases in patients who have had a stroke or have episodes of transient ischemic attacks; peripheral arterial diseases.
Interaction with other drugs and other types of interactions
Increases the absorption of tetracyclines from the gastrointestinal tract, reduces the absorption of penicillins and chloramphenicol.
Lithium. With simultaneous use, diclofenac may increase the concentration of lithium in the blood plasma. It is recommended to monitor the level of lithium in the blood plasma.
Digoxin. Diclofenac may increase plasma concentrations of digoxin when administered concomitantly. It is recommended to monitor the level of digoxin in the blood plasma.
Diuretics and antihypertensive drugs. As with other NSAIDs, concomitant use of diclofenac may reduce the hypotensive effect of diuretics or antihypertensive drugs (eg, beta blockers, angiotensin-converting enzyme (ACE) inhibitors) by inhibiting the synthesis of vasodilatory prostaglandins. Therefore, this combination should be used with caution in patients, especially elderly patients, and blood pressure should be checked regularly. Patients should be adequately hydrated and renal function should be monitored at the start of combination therapy and regularly thereafter, including due to the increased risk of nephrotoxicity with diuretics and ACE inhibitors.
Anticoagulants and antithrombotic agents. It is recommended to prescribe with caution, since simultaneous use increases the risk of bleeding.
There is evidence of an increased risk of bleeding in patients taking diclofenac and anticoagulants concomitantly. Therefore, careful monitoring of patients who simultaneously use diclofenac and anticoagulants is recommended, and, if necessary, dose adjustment of anticoagulants. The drug may enhance the effect of anticoagulants, which requires monitoring of coagulation parameters during simultaneous use. Like other non-steroidal anti-inflammatory drugs, diclofenac in high doses can reversibly inhibit platelet aggregation.
Other NSAIDs, including selective cyclooxygenase-2 inhibitors and corticosteroids. Concomitant use of diclofenac and other systemic NSAIDs or corticosteroids increases the risk of gastrointestinal bleeding or ulcers. The simultaneous use of two or more NSAIDs should be avoided. Concomitant use of diclofenac and corticosteroids may increase the incidence of adverse reactions. When using the drug, the need for NSAIDs decreases.
There are reports of both hypoglycemic and hyperglycemic reactions after the use of diclofenac. For this reason, it is recommended to monitor your blood glucose levels.
Methotrexate. Diclofenac may inhibit tubular renal clearance of methotrexate, resulting in increased methotrexate levels. NSAIDs, including diclofenac, should be used with caution when prescribed less than 24 hours before treatment with methotrexate as methotrexate blood levels may rise and methotrexate may be potentiated. Cases of serious toxicity have been observed when methotrexate and NSAIDs, including diclofenac, were administered less than 24 hours apart. This interaction is mediated by the accumulation of methotrexate as a result of impaired renal excretion in the presence of NSAIDs.
Cyclosporine. Diclofenac, like other NSAIDs, may increase the nephrotoxicity of cyclosporine due to its effect on renal prostaglandins. In this regard, the drug should be used in doses lower than for patients not receiving cyclosporine. May affect blood concentrations of cyclosporine and warfarin.
Tacrolimus. The risk of nephrotoxicity is increased when NSAIDs are coadministered with tacrolimus. This may be indirect through the mechanism of renal prostaglandin suppression by both NSAIDs and calcineurin inhibitors.
Quinolone antibiotics. Through the interaction of quinolone antibiotics and NSAIDs, seizures may occur. This phenomenon may occur regardless of the patient's history of epilepsy or seizures. Therefore, quinolone antibiotics should be used with caution in patients already receiving NSAIDs.
Phenytoin. When using phenytoin and diclofenac concomitantly, the concentration of phenytoin in the blood plasma should be monitored, taking into account the expected increase in phenytoin exposure.
Colestipol and cholestyramine. These drugs may delay or reduce the absorption of diclofenac. Despite this, it is recommended to use diclofenac at least 1:00 before or 4-6 hours after the administration of colestipol / cholestyramine.
Cardiac glycosides. The simultaneous use of cardiac glycosides and NSAIDs in patients may worsen heart failure, reduce glomerular filtration rate (GFR) and increase the concentration of the glycoside in the blood plasma.
Mifepristone. NSAIDs should not be used for 8-12 days after taking mifepristone because NSAIDs may reduce the effect of mifepristone.
Potent CYP2C9 inhibitors. It is recommended to prescribe diclofenac with caution concomitantly with potent CYP2C9 inhibitors (for example, sulfinpyrazone and voriconazole), which may lead to a significant increase in the maximum plasma concentration and exposure of diclofenac due to suppression of the metabolism of diclofenac.
Selective serotonin reuptake inhibitors (SSRIs). Concomitant use of NSAIDs and SSRIs increases the risk of gastrointestinal bleeding.
Drugs, the use of which can cause hyperkalemia.
Concomitant treatment with potassium-sparing diuretics, cyclosporine, tacrolimus or trimethoprim may lead to an increase in serum potassium levels, which should be monitored.
Antidiabetic drugs. Clinical studies have shown that diclofenac can be administered concomitantly with oral hypoglycemic drugs without affecting their clinical effect. However, there have been isolated reports of hyperglycemia and hypoglycemia, which require dose adjustment of antidiabetic drugs during the use of diclofenac.
Monitoring blood glucose levels is recommended as a precaution during combination therapy.
The effectiveness of treatment increases when the diet is enriched with vitamins A, C and salts of manganese, magnesium, copper, zinc, and selenium.
Features of application
Exceed the recommended dose.
In connection with the use of selective COX-1/COX-2 inhibitors and certain NSAIDs, the risk of thrombotic cardiovascular and cerebrovascular complications increases. The simultaneous use of the drug and systemic non-steroidal anti-inflammatory drugs, such as selective cyclooxygenase-2 inhibitors, should be avoided due to the lack of any evidence of a synergistic effect and due to potential additive side effects. Caution should be exercised when prescribing treatment to elderly patients, especially debilitated patients or patients with low body weight. Like other NSAIDs, diclofenac, due to its pharmacodynamic properties, can mask the symptoms of infection. The patient's needs for diclofenac use for symptomatic relief and response to therapy should be reviewed periodically.
Cardiovascular and cerebrovascular manifestations.
Patients with a history of hypertension and/or mild to moderate congestive heart failure require appropriate medical monitoring and counseling as fluid retention and edema have been reported with the use of NSAIDs, including diclofenac. With long-term treatment, the risk of arterial thrombosis (for example, myocardial infarction or stroke) increases. In patients with uncontrolled hypertension, congestive heart failure, known coronary artery disease, peripheral arterial occlusive disease and/or cerebrovascular disease and cardiovascular risk factors (eg, hypertension, hyperlipidemia, diabetes mellitus, smoking), administer diclofenac after careful clinical evaluation. Since the cardiovascular risks of diclofenac may increase with increasing dose and duration of treatment, it should be used for the shortest possible period and at the most effective dose.
Effect on the gastrointestinal tract.
Gastrointestinal bleeding (hematemesis, ground), ulceration, or perforation, which may be fatal, has been reported with the use of nonsteroidal anti-inflammatory drugs, including diclofenac. These events may occur at any time during treatment, regardless of the presence or absence of warning symptoms or a history of serious gastrointestinal events. In elderly patients, such complications usually have more serious consequences. If gastrointestinal bleeding or ulceration occurs in patients receiving diclofenac treatment, this drug should be discontinued. As with all NSAIDs, careful medical supervision and special caution are needed when prescribing diclofenac to patients with symptoms suggestive of gastrointestinal disorders, suspected ulcers, bleeding, or perforation. The risk of bleeding, ulceration or perforation in the gastrointestinal tract increases with increasing doses of NSAIDs, including diclofenac, and in patients with a history of ulcers, particularly those complicated by bleeding or perforation. Elderly patients have an increased incidence of adverse reactions to NSAIDs, especially gastrointestinal bleeding and perforation, which can be fatal. Combination therapy with protective agents (eg misoprostol or proton pump inhibitors) should be considered for such patients, as well as for patients requiring concomitant use of drugs containing low doses of acetylsalicylic acid (ASA) / aspirin or other drugs that increase the risk for the gastrointestinal tract. Patients with a history of gastrointestinal toxicity, especially the elderly, should be monitored for unusual abdominal symptoms (especially gastrointestinal bleeding). Caution should be exercised in patients receiving concomitant treatment with drugs that increase the risk of ulceration or bleeding, such as systemic corticosteroids, anticoagulants such as warfarin, selective serotonin reuptake inhibitors (SSRIs) or antiplatelet drugs such as acetylsalicylic acid. To reduce the risk of gastrointestinal toxicity in elderly patients, treatment is initiated and maintained at low effective doses.
Effect on the liver.
Careful medical supervision is necessary if the drug is prescribed to patients with impaired liver function, as their condition may worsen. As with all NSAIDs, including diclofenac, the levels of one or more liver enzymes may increase. During long-term use of diclofenac, regular monitoring of liver function is indicated as a preventive measure. If deviations from normal liver function tests persist or worsen, clinical symptoms of liver disease appear, or other manifestations are observed (eosinophilia, rash), use of the drug should be discontinued. Hepatitis can develop when taking diclofenac without prodromal symptoms. Diclofenac should be used with caution in patients with hepatic porphyria, as it may cause exacerbation.
Effect on the kidneys.
Since fluid retention and edema have been observed with the use of NSAIDs, including diclofenac, special caution should be exercised in patients with impaired cardiac or renal function, a history of hypertension, elderly patients, patients receiving concomitant treatment with diuretics or drugs that may significantly affect renal function, as well as in patients with a significant decrease in extracellular fluid volume for any reason, for example before or after major surgery. In such cases, when using diclofenac, it is recommended to monitor renal function. After cessation of therapy, patients' condition usually returns to the state that preceded treatment.
Effect on skin.
Serious skin reactions, some fatal, have been reported, including exfoliative dermatitis, Stevens-Johnson syndrome and toxic epidermal necrolysis, which have been observed very rarely with the use of NSAIDs, including diclofenac. The highest risk of developing these reactions exists at the beginning of therapy, in most cases in the first month of treatment. You should stop taking the drug at the first appearance of skin rash, mucosal ulcers or any other manifestations of hypersensitivity.
SLE and mixed connective tissue diseases.
Patients with systemic lupus erythematosus (SLE) and mixed connective tissue diseases may be at increased risk of aseptic meningitis.
Hematological manifestations.
During long-term treatment with diclofenac, as with the use of other NSAIDs, control blood tests with determination of the number of formed elements are recommended. The drug can reversibly inhibit platelet aggregation. Patients with impaired hemostasis, bleeding diathesis or hematological abnormalities require careful monitoring.
Use in the presence of asthma.
In patients with asthma, seasonal allergic rhinitis, swelling of the nasal mucosa (ie, polyps), chronic obstructive pulmonary disease, or chronic respiratory tract infections (especially when associated with allergic rhinitis-like symptoms), reactions to NSAIDs such as exacerbations asthma (so-called aspirin asthma with analgesic intolerance), angioedema and urticaria are more common than in other patients. In such cases, special measures are recommended (preparedness for emergency care). This also applies to patients with allergic reactions to other substances, such as rash, itching, hives. As with other drugs that inhibit prostaglandin synthetase activity, diclofenac sodium and other NSAIDs may cause bronchospasm in patients with asthma, including those with a history of asthma.
Use during pregnancy and lactation
Female Fertility: Like other NSAIDs, diclofenac may affect female fertility and is therefore not recommended for women planning pregnancy. Discontinuation of diclofenac should be considered in women who are unable to become pregnant and in women undergoing fertility evaluation.
The drug should not be used during pregnancy and breastfeeding.
The ability to influence the reaction rate when driving vehicles or other mechanisms
Usually, when taking the drug in the recommended dose and with a short course of treatment, there is no effect on the rate of reactions. However, patients who experience dysfunction of the central nervous system when using the drug Movex ® Active should not drive vehicles or operate machinery.
Directions for use and doses
For adults, take 1 tablet 3 times a day. The tablets are taken orally, after meals, with water.
The duration of treatment at the recommended dose should not exceed 3 weeks.
The treatment regimen should be selected individually. If prescribed by a doctor, treatment may be longer.
The drug should be used in the most effective doses for the shortest period of time, taking into account the treatment objectives of each individual patient.
After pain relief and consultation with a doctor, treatment can be continued with Movex® Comfort.
Children
The drug should not be used in children.
Overdose
Possible increased side effects.
Symptoms: there is no typical clinical picture for an overdose of diclofenac. In case of overdose, the following symptoms may occur: headache, nausea, vomiting, epigastric pain, gastrointestinal bleeding, diarrhea, dizziness, disorientation, agitation, coma, drowsiness, ringing in the ears, fainting or convulsions. In case of severe poisoning, acute renal failure and liver damage may develop.
Treatment of acute poisoning with NSAIDs, including diclofenac, usually consists of supportive measures and symptomatic treatment of complications such as hypotension, renal failure, convulsions, gastrointestinal disorders, and respiratory depression. Carrying out special measures such as phosphorus diuresis, dialysis or hemoperfusion does not contribute to the accelerated removal of NSAIDs from the body due to the high degree of protein binding and extensive metabolism. In case of a potentially toxic overdose, it is necessary to use activated carbon; in case of a potentially life-threatening overdose, evacuate the contents of the stomach (induce vomiting, rinse the stomach).
Adverse reactions
Usually the drug is well tolerated.
Most side effects after using the drug Movex ® Active are due to the content of diclofenac and are dose-dependent.
From the immune system: hypersensitivity reactions, including itching, rash (including erythematous, bullous), urticaria, eczema, erythema multiforme (including Stevens-Johnson syndrome), toxic epidermal necrolysis, erythroderma (exfoliative dermatitis) , anaphylactic and anaphylactoid reactions (in particular hypotension and shock), angioedema, bronchial asthma (including shortness of breath), pneumonitis, vasculitis.
From the gastrointestinal tract: abdominal pain, nausea, vomiting, diarrhea, cramping sensation, dyspepsia, bloating, anorexia, stomatitis, stomatitis, glossitis, changes in the esophagus, gastritis, stomach and intestinal ulcers, including bleeding or perforation (sometimes fatal, especially in elderly patients), gastrointestinal bleeding (haematemesis, melena, bloody diarrhea), the occurrence of diaphragm-like strictures in the intestine, disorders of the lower intestine such as colitis, hemorrhagic colitis , exacerbation of ulcerative colitis or Crohn's disease, constipation, pancreatitis.
From the liver: impaired liver function, increased levels of aminotransferases in the blood serum, hepatitis, which is accompanied or not accompanied by jaundice, fulminant hepatitis, liver necrosis, liver failure.
From the nervous system: headache, dizziness, drowsiness, insomnia, sensory disturbances, including paresthesia, memory disorders, disorientation, vertigo, irritability, increased fatigue, confusion, hallucinations, cerebrovascular accident, convulsions, depression, anxiety, general weakness , nightmares, tremors, psychotic disorders, aseptic meningitis, optic neuritis.
From the senses: visual impairment (blurred vision, diplopia), hearing impairment, tinnitus, impaired taste.
Allergic reactions: hair loss, photosensitivity, purpura, including allergic purpura, dermatitis.
From the genitourinary system: edema, acute renal failure, changes in urine sediment (hematuria, proteinuria), interstitial nephritis, nephrotic syndrome, papillary necrosis, medullary necrosis of the kidney. Tulubointerstitial nephritis, fluid retention in the body, impotence.
From the hematopoietic system: anemia (including hemolytic, aplastic), thrombocytopenia, leukopenia, agranulocytosis.
From the cardiovascular system: palpitations, chest pain, arterial hypertension, heart failure. Clinical trial and epidemiological data indicate an increased risk of thrombotic complications (eg myocardial infarction or stroke) associated with the use of diclofenac, particularly at high therapeutic doses (150 mg per day) and with long-term use.
Best before date
3 years.
Storage conditions
Store out of the reach of children, in the original packaging at a temperature not exceeding 25 ° C.
Package
30 or 60 film-coated tablets per bottle, in cardboard packaging.
Vacation category
On prescription.
Manufacturer
Meditop Pharmaceutical Ltd.
Manufacturer's location and address of place of business
Hungary, Eddie Endre J. 1., Pilisborosjeno, 2097.
Applicant
Language Health GmbH.
Applicant's location
35 Jägeristrasse, Baar, 6340, Switzerland.
Compound
Glucosamine sulfate is the most important active ingredient in Movex. This component is responsible for restoration processes in bone tissue, accelerates the production of synovial fluid and prevents bone loss. The composition also contains diclofenac sodium and chondroitin sulfate.
These components are essential for maintaining healthy connective tissue. With any damage, these substances slow down in production, which causes their deficiency. A person is faced with a pain syndrome that gradually increases. To establish the regeneration of damaged tissues, therapy is carried out with Movex tablets.
Stock
active ingredients:
glucosamine sulfate, sodium chondroitin sulfate, diclofenac potassium;
1 tablet, sealed, containing glucosamine sulfate 500 mg, sodium chondroitin sulfate 400 mg, potassium diclofenac 50 mg;
additional speeches:
povidone, microcrystalline cellulose, anhydrous silica, talc, croscarmellose sodium, magnesium stearate, sodium starch glycolate (type A), hypromelose, polyethylene glycol 6000, titanium dioxide (E 171), barv nik zhovty zakhid FCF (E 110).
Cost and release form
Movex is a fairly powerful drug that is highly popular among patients. It is usually prescribed to slow down degenerative processes in tissues. The product is produced by an Indian company in two forms:
- Movex Active - tablets of 30 or 60 pieces, the average cost of which is 1000-1600 Russian rubles;
- Movex Comfort - tablets of 30 or 60 pieces, which can be purchased for 800 or 1400 Russian rubles.
Indications for use
Movex is a drug that represents a group of chondroprotectors. It is designed to protect cartilage tissue from destruction. The product has a combined effect and has a positive effect on the functioning of the entire body.
High efficiency from the use of the drug is ensured due to its unique composition. The medicine is based on glucosamine and chondroitin. In a mutual reaction, the components of the active substance react with each other, due to which the cartilage tissue is restored in a person.
Movex’s action is also aimed at normalizing the chemical properties of synovial fluid.
The active components ensure that it constantly maintains a viscous, dense consistency. They reduce the activity of elastase and hyaluronidase, which have a destructive effect on the structure of cartilage tissue.
With an integrated approach, you will be able to normalize calcium-phosphorus metabolism, thereby stopping the process of bone loss. This helps slow down destructive processes, a person gets rid of inflammation and can lead a full life.
Movex is usually used in the following cases:
- Subsequently, injuries, intense physical activity that caused damage to bone or cartilage tissue;
- For pathological processes in bones: osteopathic dysfunction, osteochondropathy, chondromalacia, periarthritis;
- With degenerative changes in the spinal column or joints;
- To significantly accelerate the rate of healing of fractures;
- For the prevention of primary and secondary osteoarthritis, osteochondrosis, spondyloarthrosis.
Side effects
Make sure the drug is well tolerated.
Most side effects after taking Movex®
The active dose is used instead of diclofenac and dose-dependent.
On the side of the immune system:
hypersensitivity reactions, including itching, rash (including erythematous, bullous), urticaria, eczema, erythema multiforme (including Stevens-Johnson syndrome), toxic epidermal necrolysis, erythroderma (exfoliative dermatitis), anaphylactic anaphylactoid reactions (terminal hypotension and shock), angioedema, bronchial asthma (including shortness of breath), pneumonitis, vasculitis.
On the side of the scolio-intestinal tract:
abdominal pain, nausea, vomiting, diarrhea, cramps, dyspepsia, bloating, anorexia, stomatitis, aphthous stomatitis, glossitis, side changes, gastritis, varicose veins and intestines, including bleeding whose or perforation (sometimes with lethal legacy , especially in patients of the summer age), scolial-intestinal bleeding (vomiting with blood vessels, melena, diarrhea with blood cells), diaphragm-like strictures in the intestine, damage to the side of the lower veins in the intestines, such as colitis, nonspecific hemorrhagic colitis, acute nonspecific viral infection colitis or Crohn's disease, constipation, pancreatitis.
On the side of the liver:
impaired liver function, elevated levels of aminotransferases in blood serum, hepatitis, pancreatitis, fulminant hepatitis, liver necrosis, liver failure.
In case of stenosis of the scutulo-intestinal tract, peritonitis and ischemic colitis are more specific/severe forms of side effects on the side of the scutulo-intestinal tract.
On the side of the nervous system:
headache, confusion, drowsiness, insomnia, impaired sensitivity, including paresthesia, memory disorders, disorientation, vertigo, irritability, increased sleepiness, confusion, hallucinations, confusion cerebral hemorrhage, seizures, depression, anxiety, mental weakness, nighttime cravings , tremor, psychotic disorders, aseptic meningitis, neuritis of the visual nerve.
From the side of the organs one can feel:
impaired vision (blurred vision, diplopia), impaired hearing, noise in the ears, impaired sense of taste.
Dermatological reactions:
hair loss, photosensitivity, purpura, including allergic purpura, dermatitis.
On the side of the sechostate system:
swellings, gastrointestinal deficiency, hematuria, proteinuria, interstitial nephritis, nephrotic syndrome, papillary necrosis, medullary necrosis, impotence.
On the side of the hematopoietic system:
anemia (including hemolytic, aplastic), thrombocytopenia, leukopenia, agranulocytosis.
On the side of the cardiovascular system:
including heart palpitations, chest pain, arterial hypertension, heart failure, myocardial infarction, hypotension, vasculitis. Clinical and epidemiological data provide evidence of increased risk of thrombotic complications (for example, myocardial infarction and stroke) associated with diclofenac administration, steroids in high-dose drugs at these doses (150 mg per dose) and at trivalent congestion.
Mode of application
Using this medicine is quite simple; to understand all controversial issues, study the instructions for use in detail. The tablets are intended for oral use. They need to be washed down with plenty of water, and this does not need to be tied to meals.
During the first 3 weeks of treatment, you need to take 1-2 tablets of the drug per day; in the future, it is advisable to increase the dose of the active substance. On average, the duration of treatment is 3 months; longer treatment should be discussed with your doctor.
To achieve the best results, treatment with Movex should be carried out 2-3 times a year.
Interactions with other medicinal drugs and other types of interactions
Lithium.
With one-hour infusion, diclofenac can increase the concentration of lithium in blood plasma. It is recommended to check the blood serum level.
Digoxin.
Diclofenac may increase plasma concentrations of digoxin during one-hour stagnation. It is recommended to check the level of digoxin in blood serum.
Diuretics and antihypertensive drugs.
As with other NSAIDs, immediate administration of diclofenac may weaken the antihypertensive effect of diuretics or antihypertensive drugs (such as beta blockers, angiotensin converting enzyme (ACE) inhibitors) inhibits the synthesis of vasodilatory prostaglandins. Therefore, such a combination should be put together with caution; Patients, especially those of summer age, should regularly check their arterial pressure. Patients need to carefully remove the adhesive tissue, also monitor the function of the muscles at the beginning of the combination therapy, and then regularly, through the increased risk of nephrotoxicity when diuretics are ingested. and ACE inhibitors.
Conditions that may cause stagnation include hyperkalemia.
Concomitant treatment with potassium-sparing diuretics, cyclosporine, tacrolimus or trimethoprim may be associated with increased serum potassium levels, which should be monitored.
Other NSAIDs, including selective cyclooxygenase-2 inhibitors and corticosteroids.
Simultaneous administration of diclofenac with other systemic NSAIDs or corticosteroids may increase the incidence of adverse reactions in the gut tract. A trace of the uniqueness of the one-hour stagnation of two or more oil refineries.
Anticoagulants and antithrombotic properties.
It is recommended to administer with caution, as fragments of immediate ingestion with diclofenac may increase the risk of bleeding.
Although clinical studies do not indicate that diclofenac is added to the effect of anticoagulants, in addition to the increased risk of bleeding in patients who may immediately stagnate with diclofenac anticoagulant. It is recommended to closely monitor patients, immediately stop diclofenac and anticoagulants, and, if necessary, correct the dosage of anticoagulants. Like other non-steroidal anti-inflammatory drugs, diclofenac in high doses can reverse platelet aggregation.
Selective inhibitors of renal serotonin storage (SSRI).
Concomitant stasis of NPZD and SIZD increases the risk of bowel-intestinal bleeding.
Antidiabetic drugs.
Clinical studies have shown that diclofenac can be administered simultaneously with oral antidiabetic drugs without affecting their clinical effect. Please be aware of hyperglycemia and hypoglycemia, which will require adjustment of the dose of antidiabetic drugs during treatment with diclofenac. It is recommended to control the level of glucose in the blood as a precautionary approach during combination therapy. Possible changes in the interaction process caused by metformin/metabolic acidosis.
Methotrexate.
Use NSAIDs, including diclofenac, with caution, if less than 24 years before or after treatment with methotrexate, as blood concentrations of methotrexate may increase and increase the toxicity of methotrexate . Serious toxicity was avoided when methotrexate and NSAIDs, including diclofenac, were administered at intervals of less than 24 years. This interaction is mediated by the accumulation of methotrexate as a result of impaired nitric excretion in the presence of NSAIDs.
Cyclosporine.
Diclofenac, like other NSAIDs, may enhance the nephrotoxicity of cyclosporine through the infusion of narcotic prostaglandins. Therefore, diclofenac should be used in doses lower than that of cyclosporine for patients.
Tacrolimus.
When an NSAID is administered with tacrolimus, there is an increased risk of nephrotoxicity, which may be mediated by the suppression of nitric prostaglandins due to the NSAID and the calcineurin inhibitor.
Antibiotics of the quinolone series.
There were also reports of lawsuits that may have been related to the sudden deterioration of quinolones and NSAIDs. Such episodes are possible in patients either due to the obviousness or the presence of a history of epilepsy or trial. In connection with this, quinolone antibiotics should be used with caution in patients who are already treating NSAIDs.
Colestipol and cholestyramine.
These drugs may cause interference or changes in the administration of diclofenac. However, it is recommended to take diclofenac 1 year before or 4-6 years after taking colestipol/cholestyramine.
Cardiac glycosides.
Acute depletion of cardiac glycosides and NSAIDs may aggravate cardiac failure, alter glomerular filtration rate and increase the concentration of glycosides in blood plasma.
Mifepristone.
NSAIDs should not persist for 8-12 days after taking mifepristone; some NSAIDs may alter the effect of mifepristone.
Strong CYP2C9 inhibitors.
Care should be taken to combine diclofenac with potent CYP2C9 inhibitors (such as sulfinpirazone and voriconazole), which can lead to a significant increase in the maximum concentration in blood plasma and exposure to diclofenac over time. hunok suppressed the metabolism of diclofenac.
Phenytoin.
With one-hour administration of phenytoin and diclofenac, monitor the concentration of phenytoin in the blood plasma in order to detect increased exposure to phenytoin.
Increased absorption of tetracyclines from the scolial-intestinal tract replaces the absorption of penicillins and chloramphenicol.
Side effects
Movex is a fairly safe drug that has a positive effect on the functioning of the body. It extremely rarely causes side effects, usually due to improper use of the drug.
Among the negative consequences of using the drug are:
- Headache and dizziness;
- Nausea and vomiting;
- Lack of appetite;
- Diarrhea and constipation;
- Severe pain in the stomach area;
- The appearance of allergic reactions;
- Insomnia and nervousness;
- Impaired concentration.
Contraindications
To prevent negative consequences, it is necessary to stop using Movex if there are contraindications. The manufacturer does not recommend taking this product in the following cases:
- In case of individual intolerance to components or hypersensitivity to them;
- With developing thrombophlebitis or phenylketonuria;
- With an increased tendency to bleeding;
- For diabetes and other endocrine diseases;
- In children under 12 years of age.
To prevent side effects, read the instructions for use in advance. It is strictly forbidden to take Movex if there are contraindications.
Note!
Description of the drug Movex Active table. p/o No. 30 on this page is a simplified author’s version of the apteka911 website, created on the basis of the instructions for use.
Before purchasing or using the drug, you should consult your doctor and read the manufacturer's original instructions (attached to each package of the drug). Information about the drug is provided for informational purposes only and should not be used as a guide to self-medication. Only a doctor can decide to prescribe the drug, as well as determine the dose and methods of its use.
Overdose
If Movex is used incorrectly, a person may experience an overdose. Most often, it manifests itself in the form of an exacerbation of all side effects, but if the dose is exceeded too much, the patient develops other, more serious pathologies. These include thrombotic complications, optic neuritis, inflammatory process in the gastrointestinal tract, and the opening of internal bleeding.
To get rid of signs of overdose, you need to immediately induce vomiting to cleanse the stomach and take sorbents. If necessary, call an ambulance.
special instructions
Do not exceed the recommended dose.
Use during pregnancy and lactation. The drug is not used during pregnancy and breastfeeding.
Children. The drug is used in patients over the age of 18 years.
The ability to influence reaction speed when driving vehicles or working with other mechanisms. The drug may affect the reaction rate, so it is not recommended to use it when driving vehicles or working with complex mechanisms.
Features of application
You should not take Movex simultaneously with penicillin antibiotics - this significantly reduces their absorption, which reduces the effectiveness of such therapy. If you take the tablets together with tetracycline antibiotics, the concentration of active substances in the blood increases.
You should not take Movex together with anticoagulants or glucocorticoids - as a result of a chemical reaction, dangerous components are formed that cause abnormalities in the body's functioning.
Analogs
Movex is a fairly expensive product that is not supplied to all pharmacies. There are its complete analogues, which are no less effective. You can always replace this drug:
- Actinak 100;
- Remisid;
- Ameolin;
- Indomethacin;
- Ketonal.
Remisid
Indomethacin
Ketonal
Reviews
Vasily, 44 years old, Volgograd “When I underwent a health examination, a specialist said that I was developing osteochondrosis. I didn’t think much of it right away, but after a few months I encountered serious pain. I consulted a doctor who advised me to take a course of Movex. After only 3 months I was able to return to a full-fledged lifestyle and got rid of constant nagging pain. I recommend this product to everyone, it really works.”
Igor, 36 years old, Voronezh “Several years ago I broke my leg and could not recover for a long time. Even after complete fusion, I continued to limp due to discomfort. The doctor advised me to take Movex. I took these pills for about 3 months, but after a few weeks I felt significant relief. By the first month I was free of pain at rest, and by the end of the second month I was free of discomfort during active activities. The only thing I didn’t like was a slight heartburn after use.”
Overdose
It is possible to reduce side effects.
Symptoms:
There is no typical clinical picture with an overdose of diclofenac. In case of an overdose, the following symptoms may occur: headache, fatigue, vomiting, pain in the epigastric region, gastrointestinal bleeding, diarrhea, confusion, disorientation, awakening, coma, drowsiness, ringing in the ears, uneasiness or judge. In cases of severe illness, acute nicotine deficiency and liver damage may develop.
Likuvannya
Acute withdrawal of non-steroidal anti-inflammatory drugs, including diclofenac, requires supportive visits and symptomatic treatment of conditions such as arterial hypotension, nitric deficiency, vascular disease, damage to the side of the colon. kovy tract, depressed diet. Carrying out special treatments, such as phosphorus diuresis, dialysis or hemoperfusion, does not facilitate the rapid elimination of NSAIDs from the body due to the high level of protein binding and extensive metabolism. In cases of potentially toxic overdose, it is necessary to stagnate the active vugill; In case of potentially unsafe overdose for life, evacuate instead of the tube (click vomit, flush the tube).